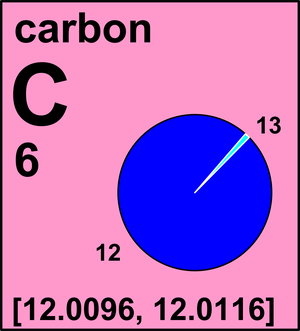
PLS HELP The average atomic mass of carbon is 12.01 amu. Based on the atomic masses of the two isotopes of - Brainly.com

C Carbon Isotopes structure chemistry Infographic - Protium, Deuterium and Tritium - chemical Useful diagram showing protons, neutrons and electrons, for education, lab, physics and science lecture. vector de Stock | Adobe Stock
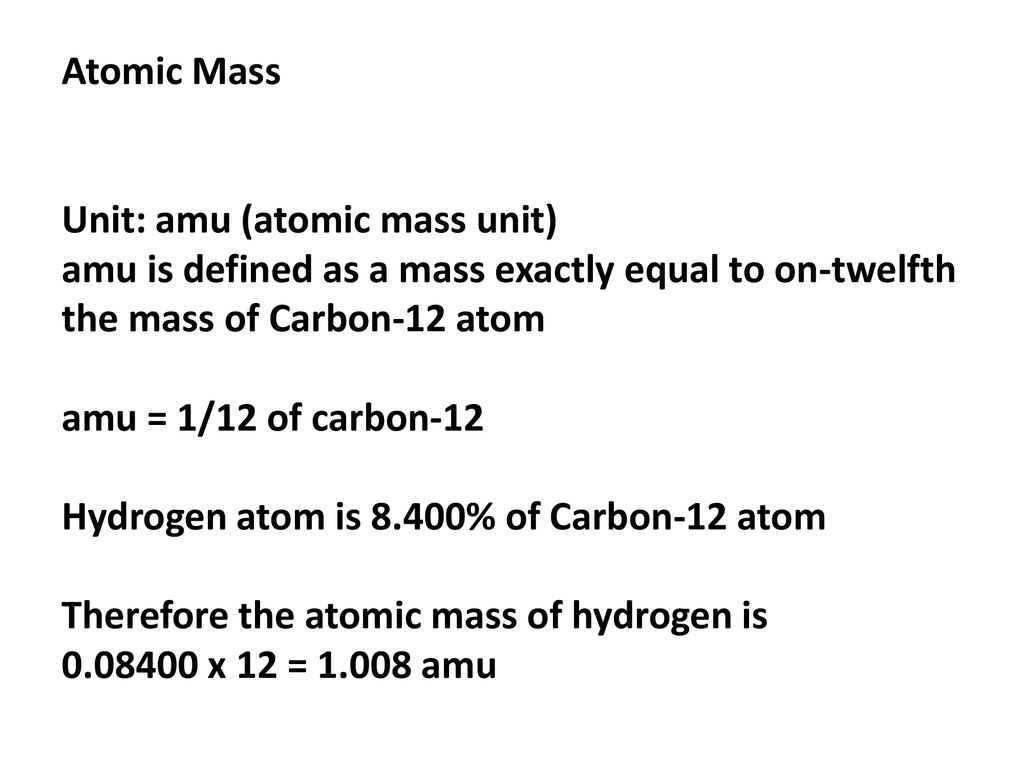
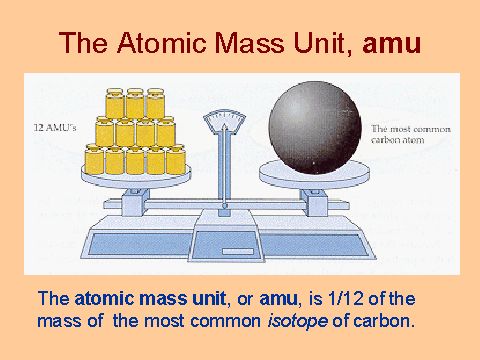
Atomic Mass Unit: amu (atomic mass unit) amu is defined as a mass exactly equal to on-twelfth the mass of Carbon-12 atom amu = 1/12 of carbon-12 Hydrogen. - ppt download

Using the Periodic Table 6 Carbon C amu Atomic number- always a whole number, increases in order, represents the number of protons in each atom. - ppt download

An atom is 10 times heavier than 112 th of mass of a carbon atom ( C - 12 ) The mass of the atom in amu is ?

AMU-Casco de fibra de carbono para motocicleta, protector de cabeza de cara abierta, estilo Retro Vintage, para Motocross - AliExpress

Atomic Mass Unit: amu (atomic mass unit) amu is defined as a mass exactly equal to on-twelfth the mass of Carbon-12 atom amu = 1/12 of carbon-12 Hydrogen. - ppt download
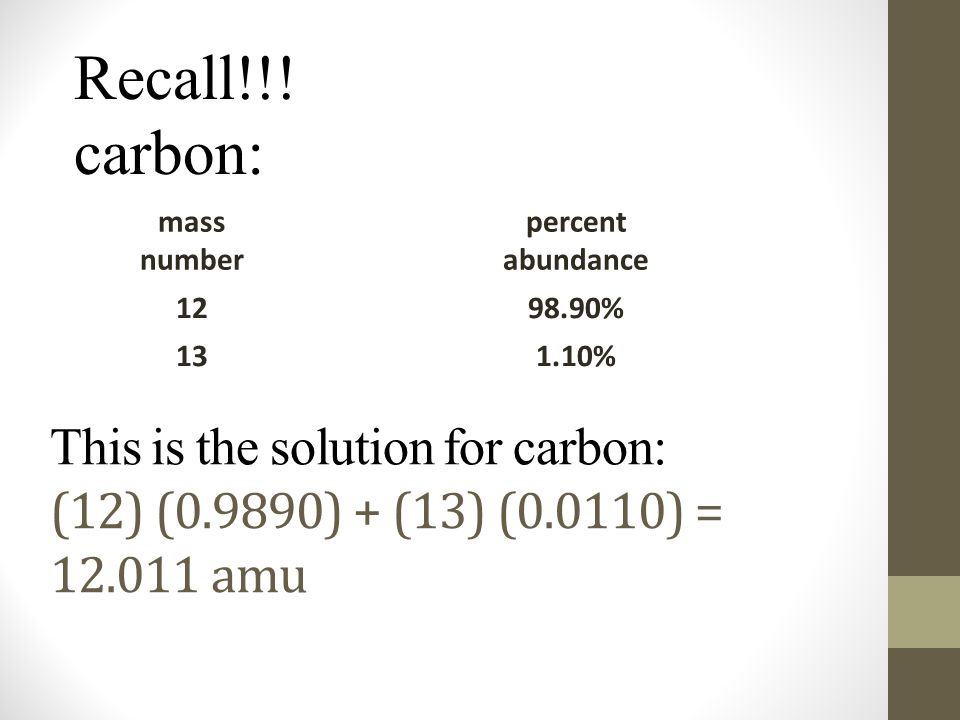
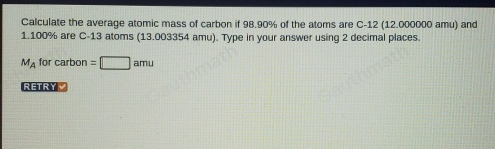
This is the solution for carbon: (12) (0.9890) + (13) (0.0110) = amu mass number percent abundance % % Recall!!! carbon: - ppt download

SOLVED: What is the total mass of carbon (in amu) in each of the following molecules? (a) CH4: 12.01 amu (b) CHBr: amu (c) C11H16O2: amu (d) CH3CH2CHCHCH2CH3: amu

If the atomic weight of carbon is set at 24 amu, the value of the avogadro constant would be :- - YouTube





![total mass (amu) of carbon [OPENSTAX] - YouTube total mass (amu) of carbon [OPENSTAX] - YouTube](https://i.ytimg.com/vi/y1I6NAv7WsI/maxresdefault.jpg)